It is one of the popular and best water filtration methods available in the market today. It forces water across a semi-permeable membrane, leaving contaminants behind that are flushed down the drain.
Table Of Contents
−- Osmosis
- Semi-Permeable Membrane
- A brief history of reverse osmosis
- How Does A Reverse Osmosis System Work?
- What Does A RO System Remove?
- Does Reverse Osmosis Wastewater?
- How to Reduce Wastewater In A RO System?
- Reverse Osmosis Performance and Design Calculations
- See the example below for more information:
- The Difference Between Passes and Stages in A Reverse Osmosis System
- Reverse Osmosis Pretreatment
- Other Pretreatment Solutions for RO Systems
In other words, this method allows you to deionize or demineralize water by forcing it under pressure through a semi-permeable reverse osmosis membrane.

Osmosis
To have a better understanding of RO, it is prudent also to understand osmosis, which is a naturally occurring phenomenon. Osmosis is a process where a weaker saline solution will tend to migrate to a strong saline solution. A good example is how our kidney absorbs water from our blood.
Semi-Permeable Membrane
This membrane allows some molecules or atoms to pass but not others. A good comparison of this membrane is a screen door. It only allows air molecules to pass through, not pests or anything larger than the holes in the screen door.
A brief history of reverse osmosis
Reverse osmosis, popularly known as RO water, is a process through which water is passed through a semi-permeable membrane. As a result, ions, molecules, and larger particles are removed (Wikipedia).
In 1748, French physicist Jean Antoine Nollet used a pig’s bladder in reverse osmosis to illustrate the process. However, nothing about the process came up until over 200 years later.
In 1949, studies from the University of California proved the effectiveness of semi-permeable membranes (RO membranes) in desalinating seawater.
Then, in the middle 1950s, researchers came up with fresh water from seawater. In addition, scientists proved that the method was viable for producing pure water.
However, the cost and technology for producing the membrane were still a setback. Finally, John Cadotte made a breakthrough, and by 2001, over 15,000 desalination plants were distributed worldwide. Click on the video below to see water desalination using RO plants:
Today, over 80% of desalination plants use the RO process. As a result, you can buy an RO filtration plant for your home today. It has proven to be still one of the most effective water filtration methods, and it is cost-effective.
It is an excellent system to satisfy the growing population’s daily rising need for clean water. Moreover, reverse osmosis is useful in cleaning water from various sources; hence, very effective, considering that most water sources are already polluted.
Manufacturers like APEC guaranteed consumers that their RO systems purify water up to 99%. This is the best relief for safe drinking water. Find the reverse osmosis system buyers guide here and be able to choose the perfect one for your home uses.
How Does A Reverse Osmosis System Work?
As mentioned above, reverse osmosis works by forcing water through a semi-permeable membrane. High pressure is used to increase pressures on the salt side of the RO, forcing the water across the semi-permeable membrane, leaving contaminants behind that are then flushed down the drain. You are finally left with clean-tasting drink water.
What Does A RO System Remove?
RO is capable of removing almost all contaminants available in the water. That is, up to 99%+ of the dissolved salts (ions), particles, colloids, organics, bacteria, and pyrogens from the feed water.
But remember, an RO system is unreliable for removing bacteria and viruses from water. Nevertheless, RO effectively treats the surface, brackish, and groundwater for small and large flow applications.
Here is a list of impurities RO removes from water:
- Pesticides and herbicides
- Sediment
- Arsenic
- Lead
- Fluoride
- Chlorine and chloramine
- Detergents
- Nitrates & Sulfates
Benefits of A RO System
- Reduces or removes bad taste and odors.
- Reduce sodium.
- Easy to install and maintain
- Remove harmful dissolved contaminants in water.
- More environmentally friendly than bottled water
- Fits under the kitchen sink
Does Reverse Osmosis Wastewater?
An RO system sends water with rejected contaminants (removed salts, dissolved pollutants, and minerals) down the drain as wastewater.
For example, 4 gallons of water exit the drain for every gallon produced. The wastewater helps clean the water, just like a washing machine uses water to clean clothes.
How to Reduce Wastewater In A RO System?
Consider doing the following:
- Using the RO reject water for artificial lakes or landscaping.
- Add a permeate pump to a reverse osmosis system to increase its efficiency. Permeate pumps reduce the wastewater from an RO system by 75 to 80%.
- Choose a reverse osmosis system with an automatic shut-off valve. An ASO valve stops water flow to the drain once the storage tank is full.
Reverse Osmosis Performance and Design Calculations
There is a variety of calculations that are used to determine the performance and design of a reverse osmosis system. Here is what you need to measure the performance of an RO system accurately:
- Temperature
- Feed flow
- Feed pressure
- Permeate pressure
- Concreate pressure
- Feed conductivity
- Permeate flow
- Permeate conductivity
Salt Rejection %
| Salt Rejection % = | The conductivity of Feed Water – Conductivity of Permeate Water | × 100 |
| Conductivity of Feed |
The above equation tells how effective the RO membrane is at removing contaminants. In other words, this equation tells you more about how the entire system is performing rather than telling you how each membrane is performing.
For example, a well-designed RO system with properly functioning RO membranes will reject 95% to 99% of most feed water contaminants.
Note: Higher salt rejection means the system is performing well. Low salt rejection implies that the membrane requires cleaning or replacement.
Salt Passage %
The salt passage is the inverse of salt rejection. It expresses the amount of salt in percentage passing through the RO system. Here is the equation:
Salt Passage % = (1 – Salt Rejection %)
Note: Low salt percentage is an indicator the system is performing well. A higher percentage means you need to clean or replace the membrane.
Recovery %
It is the amount of water that is recovered as good permeate water or water that is not sent to drain as a concentrate.
| % Recovery = | Permeate Flow Rate (GPM) | × 100 |
| Feed Flow Rate (GPM) |
A higher recovery percentage means you send less water to drain as a concentrate, saving more permeate water. But too high a recovery percentage can lead to larger problems due to fouling and scaling.
Note: Proper recovery % at which an RO operate depends on what the RO system is designed to do. Calculating the percentage recovery can quickly determine if an RO system operates outside the intended design.
See the example below for more information:
If the recovery rate is 80%, then this means that for every 100 gallons of feed water that enter the RO system, you are recovering 80 gallons as usable permeate water, and 20 gallons will drain as a concentrate.
Concentration Factor
A crucial factor in designing an RO system, the concentration factor, is related to the RO system recovery. The more water you recover as permeate, the more concentrated salts and contaminants you collect in the concentrate stream.
This can lead to a higher potential for scaling on the surface of the RO membrane when the concentration factor is too high for the system design and feed water composition.
| Concentration Factor = | 1 |
| 1 – Recovery % |
See the example below:
If your feed flow is 100 GPM and your permeate flow is 75 GPM, then the recovery is (75/100) x 100 = 75%.
Concentration factor
1 ÷ (1-75%) = 4.
From the example, a concentration factor of 4 means that the water going to the concentrate stream will be four times more concentrated than the feed water. So, if the feed water in the above example were 600 ppm, then the concentrate stream would be 600 * 4 = 2,400 ppm.
Flux
Flux is expressed as volume per area per unit of time. Flux expresses the rate at which water permeates a reverse osmosis membrane. Typical units of measurement are gallons per square foot per day (i.e., GFD or GSFD) or liters per square meter per hour (l/m2/hr).
| Gfd = | GPM of permeate × 1,440 min/day |
| # of RO elements in system × square footage of each RO element |
Mass Balance
(Feed flow1 x Feed Conductivity) = (Permeate Flow x Permeate Conductivity)
+ (Concentrate Flow x Concentrate Conductivity)
1Feed Flow equals Permeate Flow + Concentrate Flow
The above equation determines if your flow and quality instrument is reading correctly or requires calibration. If the instrumentation is not reading correctly, that indicates that the performance data you are collecting is useless.
Data to collect from an RO system to perform mass balance calculation:
- Feed Flow (GPM)
- Permeate flow (GPM)
- Concentrate Flow (GPM)
- Feed Conductivity (µS)
- Permeate conductivity (µS)
- Concentrate Conductivity (µS)
See the example below:
| Permeate flow | 5 GPM |
| Feed conductivity | 500 µS |
| Permeate Conductivity | 10 µS |
| Concentrate Flow | 2 GPM |
| Concentrate Conductivity | 1200 µS |
The solution would be:
(7 x 500) = (5 x 10) + (2 x 1200)
3,500 ≠ 2,450
Then find the difference
(Difference / Sum) x 100
((3,500 – 2,450) / (3,500 + 2,450)) x 100
= 18%
A difference of +/- 5% is ok. A difference of +/- 5% to 10% is adequate. A difference of > +/- 10% is not acceptable, and calibration of the RO instrumentation is necessary to ensure that you are getting reliable data. From the example above, the RO mass balance equation falls out of range and requires attention.
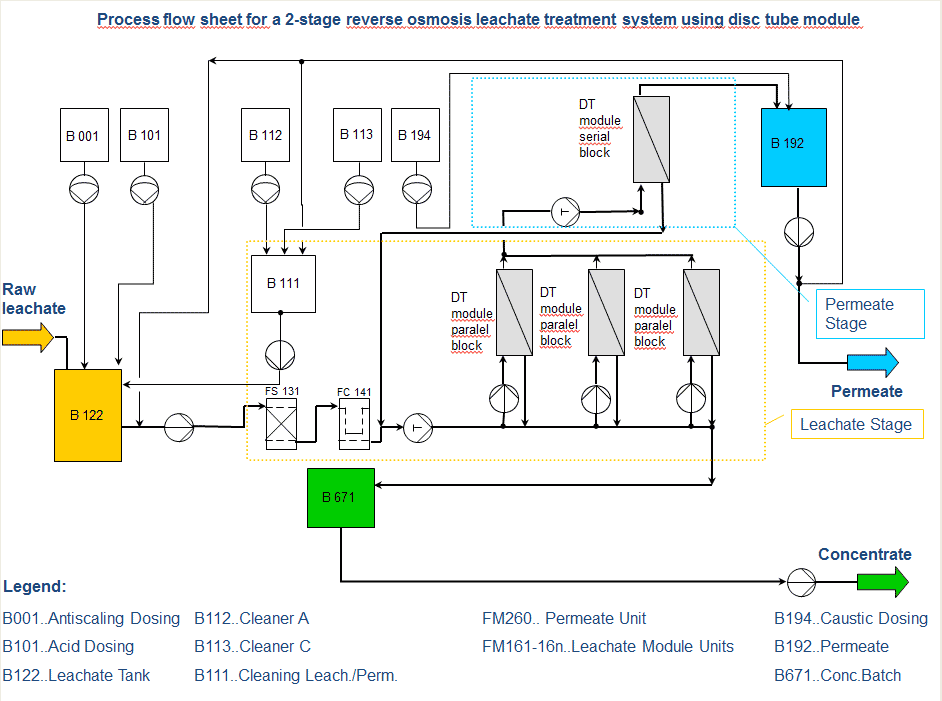
The Difference Between Passes and Stages in A Reverse Osmosis System
These two terms (pass and stage) are often mistaken for the same thing in an RO system. It cannot be very clear if you don’t get the difference. Let’s now dive deeper.
What is the difference between a 1 and 2-stage RO and a 1 and 2-pass RO
- A 1 And 2 Stage RO System
In a one-stage Ro system, the feed water enters the RO system as one stream and exits the RO as either concentrated or permeate water. While in the two-stage system, the concentration from the first stage becomes the water in the second stage.
The permeate water collected from the first stage is then combined with permeate water from the second stage. These additional stages increase the recovery from the system. An RO system with concentrate recycling can be utilized at the first stage to help improve the system recovery.
- Single Pass RO Vs. Double Pass RO
The difference between a single pass RO system and a double pass RO system is clear. When it comes to double pass RO, the permeate from the first pass becomes the feed water to the second pass, producing a much higher quality permeate because it has essentially gone through two RO systems.
A double pass also offers the opportunity to remove carbon dioxide gas from the permeate by injecting caustic between the first and second pass.
Reverse Osmosis Pretreatment
Common problems an RO system experiences are due to lack of proper pretreatment.
- Fouling
The membrane fouling is due to the emulsified or suspended materials in the feed water to the RO system. Fouling occurs when contaminants accumulate on the membrane surface, effectively plugging the membrane. Examples of such materials are the clay, silica, oil, iron, sulfur, and humic acids. These substances can be present in a very fine or colloidal form.
Proper pretreatment minimizes the need to worry about fouling-related problems. For example, multi-media filters (MMF) or microfiltration (MF) methods are commonly used to prevent fouling. However, in some cases, cartridge filtration will do.
- Scaling
Scaling causes a higher energy use and a shorter life span of the membranes. This is because you have to clean the membrane often. Scaling is the deposition of particles on a membrane during RO, causing it to plug.
Calcium carbonate is a good example of a common scale that forms on an RO membrane. The best way to prevent scaling is by using antiscalants and scale inhibitors. These are chemicals that, once added to water, reduce the scaling potential of the feed water.
Adding them increases the solubility limits of troublesome inorganic compounds, making it possible to achieve a higher recovery rate and run at a higher concentration factor.
The other option is using a water softener. A water softener can help prevent scaling in an RO system by exchanging scale-forming ions with non-scale forming ions. It is crucial to have a 5-micron cartridge filter placed directly after the water softener just in case the underdrains of the softener fail.
- Chemical attack
Modern thin-film composite membranes are not tolerant of chlorine or chloramines, and the result of any chemical attack is significant. For example, oxidizers such as chlorine cause irreparable damage by leaving huge holes in the RO membrane. In addition, such an attack results in a higher permeate flow and poor quality permeate water.
- Mechanical Damage
Various reasons can lead to mechanical damage, such as pre and post-plumbing. The best fix is using variable frequency drive motors to start high-pressure pumps for RO systems and installing check valve(s) and pressure relief valves to prevent excessive back pressure on the RO unit that can cause permanent membrane damage.
Other Pretreatment Solutions for RO Systems
- Sodium Bisulfite (SBS) Injection: SBS, a reducer once added to water stream before an RO at the right amount, can help remove residue chlorine.
- Granular Activated Carbon (GAC): GAC, which is made from coal, nutshell, or wood, is used for both removing organic constituents and residual disinfectants (such as chlorine and chloramines) from water.
RO Data Trending and Normalization
It is good to note that RO membranes are the heart of the RO system, and certain data points must be collected to determine their health. These data points include flows, temperature, and system pressures.
These data points, once analyzed, help in normalization, so those flow variations are not interpreted as abnormal when no problem exists. The analysis results also help determine the right time to clean or replace the membrane.
As a general rule of thumb, you must take action when the normalized change is +/- 15% from the baseline data. If you don’t follow this rule, RO membrane cleanings may not effectively bring the membranes back to near-new performance.
RO Membrane Cleaning
Periodic cleaning is very crucial. Feed water quality will often help you determine when to clean the RO membrane.
As a general rule, if the normalized pressure drops or the normalized salt passage has increased by 15%, it is time to clean the RO membranes. Likewise, if the normalized permeate flow has decreased by 15%, it is also time to clean the RO membranes.

Jay
Jay is a health and wellness enthusiast with expertise in water quality and nutrition. As a knowledgeable advocate for holistic well-being, Jay successfully manages Type 2 Diabetes through informed lifestyle choices. Committed to sharing reliable and authoritative insights, Jay combines firsthand experience with a passion for enhancing health."

